- Instructions: On a sheet of paper, answer all 30 questions in sentences. Due Wednesday, 10/12/16.
- Check the number near the images immediately below to see to which question it refers.
Questions
- Name the characteristics of metals.
- Based on the numbers of subatomic particles, which of the following atoms will have a positive charge?
A Protons=6, Neutrons=6, Electrons =6
B Protons=7, Neutrons=6, Electrons =6
C Protons=9, Neutrons=10, Electrons =9
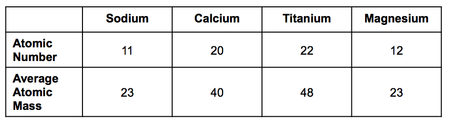
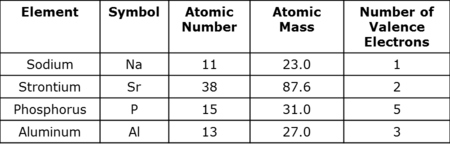
D Protons=10, Neutrons=11, Electrons =11 - A table with information about four elements is provided. Which element contains 20 protons? Which element contains 26 neutrons?
- What influences the reactivity of an element?
- Describe a proton.
- An atom of an element has 12 protons. How many electrons does the atom have if it has a charge of positive 2 (+2)?
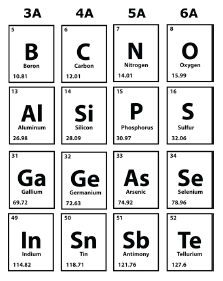
- A portion of the Periodic Table is shown. Which element would be expected to have a reactivity similar to bromine?
- A researcher wants to experiment with an element that reacts like bromine (Br) but has a greater atomic mass. Name two elements the researcher could select for the experiment?
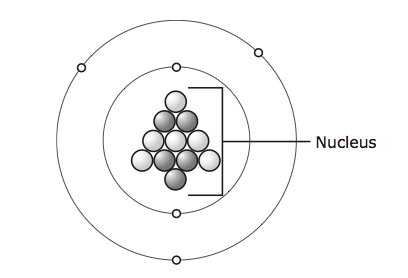
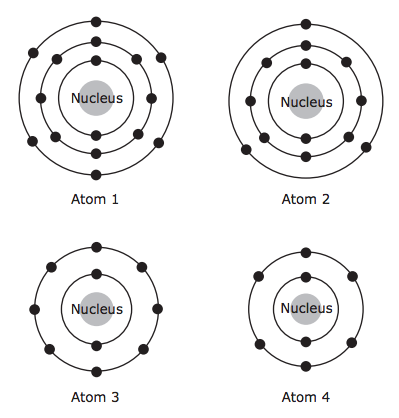
- A model of an atom is shown below.
An atom of which element is represented by this model? - When trying to identify an unknown element, a scientist determines what other elements the unknown element reacts with chemically. Which property of the unknown element determines the other elements it reacts with?
- An element can be identified by its unique number of what?
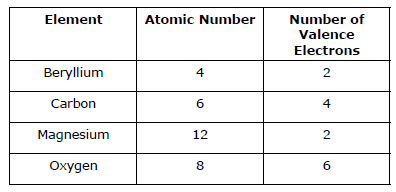
- The table provides information on four different elements. Which element has eight protons in its nucleus?
- What property uniquely identifies every
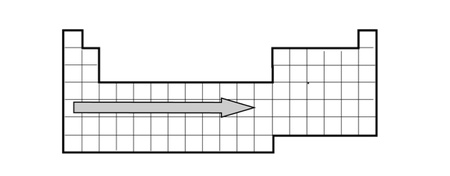
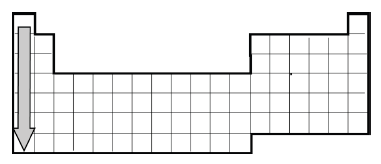
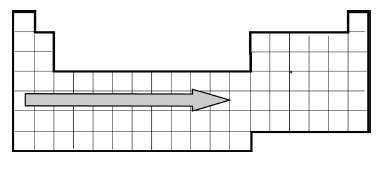
element? - A simplified version of the Periodic Table of the Elements is provided.
When moving across a row in the Periodic Table, what increases? - Provided is a representation of the Periodic Table of
Elements. What trend can be observed when moving down a column in the Periodic Table? - A portion of the periodic table is shown. Which pair of elements would have the same number of valence electrons?
- An element’s reactivity and other chemical properties are
determined by the number of what? - Molybdenum has an atomic number of 42. What does this mean?
- What property of technetium distinguishes it from all other elements?
- The table provides information on four different elements. Which is the most reactive?
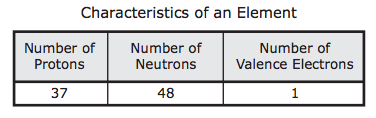
- The table below lists three characteristics of an atom of an element. An atom of which element is described by the data in the table?
- A student is studying the ways different elements are similar to one another. Diagrams of atoms from four different elements are shown below. Which two atoms are of elements in the same group in the periodic table?
- A student is studying calcium, a highly reactive element that humans need for strong bones. Whiat characteristic of calcium is most closely related to its chemical reactivity?
- Name any two elements on the periodic table that are in the same period?
- The periodic table is organized into groups and periods of elements. The characteristics of a certain group of elements are listed below. Name all the elements that are in the same group.
- The atoms of a certain element each contain 11 protons and 1 valence electron. Write a statement that correctly identifies this element and describes its chemical reactivity.
- The diagram represents part of the Periodic Table. Which pair of elements has the same number of valence electrons?
- A portion of the Periodic Table is provided. Which of the following sequences of elements would all have four valence electrons? Write four different sequences of elements that have four valence electrons.
- Provided is a representation of the Periodic Table of
Elements. When moving across a row in the Periodic Table, what trend appears? - The number of valence electrons in an atom determines what?